After 25 years of research, a big breakthrough.
By Dileep Thekkethil
BANGALORE: A team of scientists at the University of Illinois has solved the mystery behind the antibiotic agent that kills microorganisms or inhibits their growth.
Scientists relied on a few dozens of compounds that included antibiotic properties for conducting the research. Nisin, a polycyclic antibacterial peptide that is most common in milk was one among the compounds used for the research as it can be synthesised in the lab. Nisin is used by food manufacturers as a preservative to fight the food-borne pathogens. The abstract of the research says, “nisin contains dehydroalanine and dehydrobutyrine residues that are formed by the dehydration of Ser/Thr by the lantibiotic dehydratase NisB.”
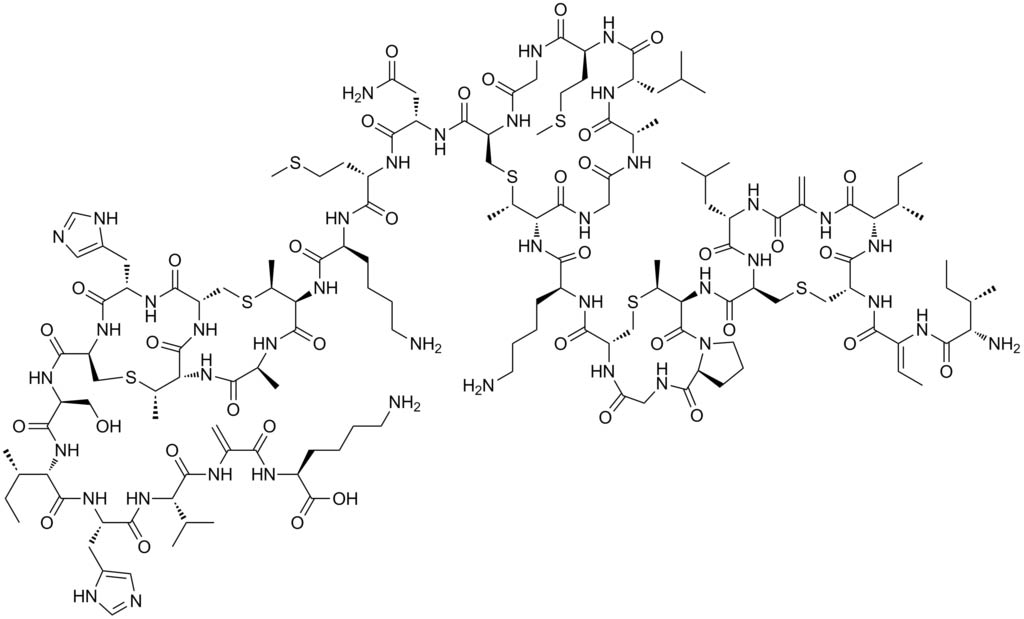
The molecular sequence of nisin gene was already known to researchers and they had the knowledge about the amino acids or peptides that are encoded by this gene. But researchers have for the last 25 years tried to understand the several transformations that the cells made before getting the final shape and function.
In the case of nisin, it carries an enzyme called dehydratase, which can remove the water to give the antibiotic the three-dimensional shape. According to Wilfred van der Donk, the lead researcher, this was the important step in transforming the spaghetti-like peptide into a five-ringed structure.
Manuel Ortega, a graduate student in van der Donk’s lab, through a careful process of elimination, established that the amino acid glutamate was essential to nisin’s transformation.
According to the biochemistry professor Satish K. Nair who assisted Prof. Donk “Ortega discovered that the dehydratase did two things. One is that it added glutamate (to the nisin peptide), and the second thing it did was it eliminated glutamate. But how does one enzyme have two different activities?”
The researchers also made the ground breaking discovery of transfer-RNA, which is a molecule that deals with protein production, supplies the glutamate, allowing the dehydratase to give nisin its final shape and function.
Prof. van der Donk said “In this study, we solve a lot of questions that people have had about how dehydration works on a chemical level. And it turns out that in nature a fairly large number of natural products – many of them with therapeutic potential – are made in a similar fashion. This really is like turning on a light where it was dark before, and now we and other labs can do all kinds of things that we couldn’t do previously.”
The study is published in the journal Nature.



