Headquartered in in Richmond, VA, Indivior Inc employs more than 900 individuals globally and its portfolio of products is available in over 40 countries worldwide.
Headquartered in in Richmond, VA, Indivior Inc employs more than 900 individuals globally and its portfolio of products is available in over 40 countries worldwide.
Indivior Inc., the US subsidiary of the British pharmaceutical company Indivior PLC, has filed a patent infringement lawsuit against Indian pharma firm Dr. Reddy’s and four other firms for their proposed generic versions of the Sublingual Film product used to treat opioid addiction.
According to the lawsuit filed by Indivior, the companies including Dr. Reddy’s are making generic versions of its SUBOXONE ®, violating the US patent law as the drug, which is an opioid used to treat opioid addiction, acute pain, and chronic pain has been patented under the United States Patent No. 9,931,305 (the ‘305 patent).
Shaun Thaxter, CEO of Indivior, said, “The ‘305 patent, along with the other new Orange Book patents that we secured (the ‘454 and ‘221 patents), demonstrates our commitment to innovate and generate new intellectual property for the SUBOXONE® Film. As a follow-on to our ‘514 patent, we believe this new patent is strong and we are asserting it to protect our intellectual property rights.â€
The lawsuit alleges that the companies including Dr. Reddy’s infringe the ‘305 patent, which was issued on April 3 and has been submitted for listing in the FDA’s Orange Book.
The ‘305 patent claims film compositions having a substantially uniform distribution of the active drug. Indivior has been involved in patent infringement litigation involving other, earlier issued patents covering SUBOXONE® Film with these same companies relating to their proposed generic products.
Headquartered in in Richmond, VA, Indivior Inc employs more than 900 individuals globally and its portfolio of products is available in over 40 countries worldwide.
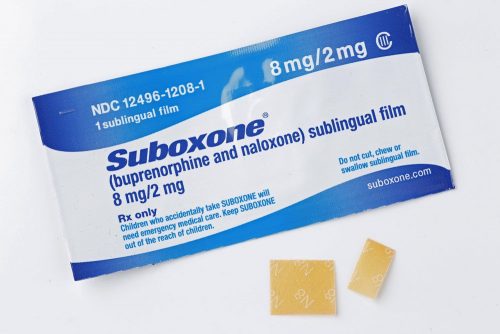
SUBOXONE® (buprenorphine and naloxone) Sublingual Film (CIII) is a prescription medicine indicated for the treatment of opioid dependence and is used as part of a complete treatment plan to include counseling and psychosocial support.
The other pharma companies mentioned in the lawsuit for apparent violation of patent rules include Actavis, Par, Alvogen and Teva.
Also Read
Dr. Reddy’s Laboratories record decline of 60.12% in second quarter
October 25, 2016
Drop in sales in Venezuela and North America.
Kallam Anji Reddy, founder of Dr. Reddy’s Laboratories, dies
March 16, 2013
Reddy, 72, was battling cancer.
Indian pharma majors under investigation for multistate antitrust scheme
November 6, 2017
Two Indian American senior executives also face lawsuit.